Difference between revisions of "Iconic symbols"
(Created page with "{{MitoPedia |abbr= |description='''Iconic symbols''' are used in ergodynamics to indicate more explicitely than standard SI or IUPAC symbols the quantity represented, part...") |
|||
| Line 1: | Line 1: | ||
{{MitoPedia | {{MitoPedia | ||
|abbr= | |abbr= | ||
|description='''Iconic symbols''' are used in [[ergodynamics]] to indicate more explicitely | |description='''Iconic symbols''' are used in [[ergodynamics]] to indicate more explicitely — compared to standard SI or IUPAC symbols — the quantity represented. This is particularly the case in normalized quantities (ratios of quantities). Iconic (or canonical) symbols help to clarify the meaning, are based on SI and IUPAC symbols as far as possible, and may be translated into more commonly used, practical symbols. Several ambiguities in SI and IUPAC symbols are eliminated by the systematic structure of iconic symbols, but it may be impossible to avoid all ambiguities, particulary when long (canonical) symbols are abbreviated in a particular context. Clarity is further improved by showing the unit of a quantity together with the iconic symbol of the quantity. | ||
|info=[[Gnaiger 2020 MitoPathways]] | |info=[[Gnaiger 2020 MitoPathways]] | ||
}} | }} | ||
__TOC__ | __TOC__ | ||
Communicated by [[Gnaiger E]] 2020-11-29 | Communicated by [[Gnaiger E]] 2020-11-29 | ||
== Abbreviation of iconic symbols == | |||
::: Exmaple | |||
::::* The symbol Δ<sub>m</sub>''F''<sub>''<u>n</u>''H<sup>+</sup></sub> [kJ·mol<sup>-1</sup>] for the ''pmF'' includes the specification of the molar format ''<u>n</u>''. If all quantities are expressed in the molar format in a particular context, then the symbol may be abbreviated as Δ<sub>m</sub>''F''<sub>H<sup>+</sup></sub> [kJ·mol<sup>-1</sup>]. This opens up an ambiguity, since in another context the same abbreviated symbol Δ<sub>m</sub>''F''<sub>H<sup>+</sup></sub> would indicate the ''pmF'' in the electrical format ''<u>e</u>'' with electrical units [V]. | |||
::::* Even the symbol Δ<sub>m</sub>''F''<sub>''<u>n</u>''H<sup>+</sup></sub> is ambiguous, since the compartmental direction of the ''pmF'' is not specified. A positive or negative numerical value of Δ<sub>m</sub>''F''<sub>''<u>n</u>''H<sup>+</sup></sub>, therefore, cannot be interpreted without further specification. This is achieved by canonical extension of the iconic symbol as Δ<sub>m</sub>''F''<sub>''<u>n</u>''H<sup>+</sup>pos</sub>, which indicates the direction from the negative compartment (matrix) to the positive compartment across the mtIM. | |||
:::: Abbreviation of iconic symbols in a well-defined context improves readability and shortens impractically long equations. | |||
== Normalized quantities == | == Normalized quantities == | ||
::: Exmaples | |||
::::* [[protonmotive force]] Δ<sub>m</sub>''F''<sub>''<u>e</u>''H+</sub> = ''pmF'' [V] | |||
::::* [[elementary charge |proton charge]] ''Q''<sub><u>''U''</u>H<sup>+</sup></sub> = ''e'' [C·x<sup>-1</sup>] | |||
::::* [[particle charge]] ''Q<sub><u>U</u>X</sub>'' [C·x<sup>-1</sup>] | |||
:::: Iconic symbols show the quantity, the format of the normalization in the subscript (''<u>U</u>'', ''<u>n</u>'', ''<u>e</u>''), and the entity specified in the subscript (''X''). The normalized quantities are '''per''' ''X''. In the quantities ''Q''<sub>el''X''</sub>, ''N<sub>X</sub>'', ''n<sub>X</sub>'', ''V<sub>X</sub>'', ''m<sub>X</sub>'', the subscript ''X'' without attachment to a format indicates the quantity '''of''' ''X''. | :::: Iconic symbols show the quantity, the format of the normalization in the subscript (''<u>U</u>'', ''<u>n</u>'', ''<u>e</u>''), and the entity specified in the subscript (''X''). The normalized quantities are '''per''' ''X''. In the quantities ''Q''<sub>el''X''</sub>, ''N<sub>X</sub>'', ''n<sub>X</sub>'', ''V<sub>X</sub>'', ''m<sub>X</sub>'', the subscript ''X'' without attachment to a format indicates the quantity '''of''' ''X''. | ||
{{Template:Keywords: Charge}} | {{Template:Keywords: Charge}} | ||
Revision as of 14:10, 29 November 2020
Description
Iconic symbols are used in ergodynamics to indicate more explicitely — compared to standard SI or IUPAC symbols — the quantity represented. This is particularly the case in normalized quantities (ratios of quantities). Iconic (or canonical) symbols help to clarify the meaning, are based on SI and IUPAC symbols as far as possible, and may be translated into more commonly used, practical symbols. Several ambiguities in SI and IUPAC symbols are eliminated by the systematic structure of iconic symbols, but it may be impossible to avoid all ambiguities, particulary when long (canonical) symbols are abbreviated in a particular context. Clarity is further improved by showing the unit of a quantity together with the iconic symbol of the quantity.
Reference: Gnaiger 2020 MitoPathways
Communicated by Gnaiger E 2020-11-29
Abbreviation of iconic symbols
- Exmaple
- The symbol ΔmFnH+ [kJ·mol-1] for the pmF includes the specification of the molar format n. If all quantities are expressed in the molar format in a particular context, then the symbol may be abbreviated as ΔmFH+ [kJ·mol-1]. This opens up an ambiguity, since in another context the same abbreviated symbol ΔmFH+ would indicate the pmF in the electrical format e with electrical units [V].
- Even the symbol ΔmFnH+ is ambiguous, since the compartmental direction of the pmF is not specified. A positive or negative numerical value of ΔmFnH+, therefore, cannot be interpreted without further specification. This is achieved by canonical extension of the iconic symbol as ΔmFnH+pos, which indicates the direction from the negative compartment (matrix) to the positive compartment across the mtIM.
- Exmaple
- Abbreviation of iconic symbols in a well-defined context improves readability and shortens impractically long equations.
Normalized quantities
- Exmaples
- protonmotive force ΔmFeH+ = pmF [V]
- proton charge QUH+ = e [C·x-1]
- particle charge QUX [C·x-1]
- Exmaples
- Iconic symbols show the quantity, the format of the normalization in the subscript (U, n, e), and the entity specified in the subscript (X). The normalized quantities are per X. In the quantities QelX, NX, nX, VX, mX, the subscript X without attachment to a format indicates the quantity of X.
Canonical comments on IUPAC definitions in the context of charge
Charge of the proton versus charge per proton
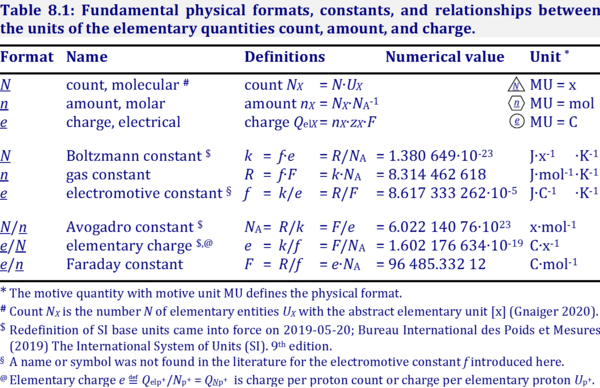
- Proton charge is the elementary charge e [C·x-1], which is charge per count of protons.
Qel ≝ Qelp+ [C]
e ≝ QNp+ = Qel·Np+-1 [C∙x-1]
- The distinction of charge of particles versus charge per single particle is not made sufficiently clear by IUPAC, when defining "-e is the charge of an electron" — it must be corrected to "-e is the charge per electron".
- For comparison, the name "charge density of electrons" is used by IUPAC with symbol ρ [C·m-3]. Dividing ρ by the count concentration of electrons [x·m-3], we obtain the unit [C·x-1] for the electron charge. Therefore, electron charge (or proton charge) is clearly the charge per particle.
Ambiguity of QB
- IUPAC (Cohen 2008 IUPAC Green Book) defines the charge number as
IUPAC: zB = QB·e-1
- Therefore, QB = zB∙e. The subscript in QB indicates per elementary entity B. This is opposite to the subscript in VB as the symbol for the volume of a substance of type B (e.g. VO2 [L]). For consistency with this convention, the symbol QelB or QelX [C] is used for indicating charge of a substance of type B or X, distinguished from particle charge as the quantity of charge per elementary entity X with symbol QNX [C∙x-1]. To avoid too long and multiple subscript levels, QNX is used instead of QUX, and the ‘el’ is dropped from QelNX. The particle charge QNH+ per hydrogen ion is identical to the definition of the elementary charge e. Therefore, the charge number of the hydrogen ion is zH+ = QNH+/e = 1. In summary:
zB = QNB·e-1
QNB = QelB·NB-1 [C∙x-1]
Keywords
- » charge QelX
- » charge number zX
- » electrochemical constant f
- » elementary charge e
- » Faraday constant F
- » hydrogen ion versus proton
- » iconic symbols
- » motive entity
- » particle charge QNX
- Bioblast links: Charge - >>>>>>> - Click on [Expand] or [Collapse] - >>>>>>>
- Normalization of charge and iconic symbols
- Iconic symbols show the quantity, the format of the normalization in the subscript (N, n, e), and the entity specified in the subscript (X). The normalized quantities are per X. In the quantities QelX, NX, nX, VX, mX, the subscript X without attachment to a format indicates the quantity of X.
Quantity Unit Normalized for quantity Unit Iconic symbol Unit Practical symbol Quantity charge QelX [C] / count NX [x] = QNX [C·x-1] particle charge (IUPAC: QB) charge QelX [C] / amount nX [mol] = QnX [C·mol-1] charge number times Faraday constant charge QelX [C] / volume VX [m3] = QVX [C·m-3] ρel charge density charge QelX [C] / mass mX [kg] = QmX [C·kg-1] specific charge count NX [x] / charge QelX [C] = NeX [x·C-1] amount nX [mol] / charge QelX [C] = neX [mol·C-1] volume VX [m3] / charge QelX [C] = VeX [m3·C-1] ρel-1 mass mX [kg] / charge QelX [C] = meX [kg·C-1]
References
- Gnaiger E (2020) Mitochondrial pathways and respiratory control. An introduction to OXPHOS analysis. 5th ed. Bioenerg Commun 2020.2. - »Bioblast link«
- Gnaiger Erich (2020) Canonical reviewer's comments on: Bureau International des Poids et Mesures (2019) The International System of Units (SI) 9th ed. MitoFit Preprint Arch 2020.4 doi:10.26124/mitofit:200004.
- Grosholz Emily R (2007) Representation and productive ambiguity in mathematics and the sciences. Oxford Univ Press 312 pp. - »Bioblast link«
MitoPedia concepts: Ergodynamics