Himms-Hagen 2001 Exp Biol Med (Maywood)
| Himms-Hagen J, Harper ME (2001) Physiological role of UCP3 may be export of fatty acids from mitochondria when fatty acid oxidation predominates: an hypothesis. Exp Biol Med (Maywood) 226:78-84. https://doi.org/10.1177/153537020122600204 |
Himms-Hagen J, Harper ME (2001) Exp Biol Med (Maywood)
Abstract: This hypothesis proposes a physiological role for uncoupling protein-3 (UCP3) in the export of fatty acid anions from muscle and brown adipose tissue (BAT) mitochondria when fatty acids are the predominant substrate being used. It proposes that excess acyl CoA within the mitochondria is hydrolyzed by a mitochondrial acyl CoA thioesterase, yielding fatty acid anion and CoASH. The fatty acid anion is exported to the cytosol by being carried across the inner mitochondrial membrane by UCP3. The CoASH is conserved within the mitochondrion to participate in other reactions for which it is needed during fatty acid oxidation in the beta-oxidation cycle and in the tricarboxylic acid cycle. The export of the fatty acid anion thus permits continued rapid fatty acid oxidation in the face of an oversupply. The hypothesis provides a logical explanation for the observed up-regulation of gene expression for UCP3 in muscle when there is a switch to fatty acid oxidation, as during fasting, and in BAT when fatty acid oxidation is stimulated, as during exposure to cold. It provides a plausible physiological role for UCP3 as a transporter protein, not as an uncoupling protein.
• Bioblast editor: Gnaiger E
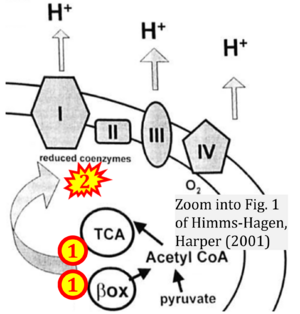
Correction: FADH2 and Complex II
- FADH2 is shown as the substrate feeding electrons into Complex II (CII). This is wrong and requires correction - for details see Gnaiger (2024).
- Gnaiger E (2024) Complex II ambiguities ― FADH2 in the electron transfer system. J Biol Chem 300:105470. https://doi.org/10.1016/j.jbc.2023.105470 - »Bioblast link«
Labels:
Tissue;cell: Skeletal muscle, Fat
Enzyme: Uncoupling protein
Pathway: F