Difference between revisions of "Arnould 2018 MiP2018b"
(Created page with "{{Abstract |title=left|90px|Mitochondrial Physiology Society|MiPsociety |info=MiP2018 |year=2018 |event=MiP2018 |abstract=Image:MITOEAGLE-lo...") |
|||
| Line 1: | Line 1: | ||
{{Abstract | {{Abstract | ||
|title=[[Image:MiPsocietyLOGO.JPG|left|90px|Mitochondrial Physiology Society|MiPsociety]] | |title=[[Image:MiPsocietyLOGO.JPG|left|90px|Mitochondrial Physiology Society|MiPsociety]] A form of autophagy triggers lipolysis in 3T3-L1 adipocytes exposed to a mitochondrial uncoupling. | ||
|info=[[MiP2018]] | |info=[[MiP2018]] | ||
|authors=Arnould T, Demine S, Tejerina S, Bihin B, Thiry M, Reddy N, Raes M, Renard P, Jadot M | |||
|year=2018 | |year=2018 | ||
|event=MiP2018 | |event=MiP2018 | ||
|abstract=[[Image:MITOEAGLE-logo.jpg|left|100px|link=http://www.mitoglobal.org/index.php/MITOEAGLE|COST Action MitoEAGLE]] | |abstract=[[Image:MITOEAGLE-logo.jpg|left|100px|link=http://www.mitoglobal.org/index.php/MITOEAGLE|COST Action MitoEAGLE]] | ||
Obesity is characterized by an excessive triacylglycerol accumulation in white adipocytes. Various mechanisms allowing the regulation of triacylglycerol storage and mobilization by lipid droplet-associated proteins as well as lipolytic enzymes have been identified. Increasing energy expenditure by inducing a mild uncoupling of mitochondria in adipocytes might represent a putative interesting anti-obesity strategy [1] as it reduces the adipose tissue triacylglycerol content by stimulating lipolysis through yet unknown mechanisms, limiting the systemic adverse effects of adipocyte hypertrophy. | |||
3T3-L1 fibroblasts were exposed to a mild uncoupling of mitochondria triggered by 0.5 μM carbonyl cyanide-p-trifluoromethoxyphenylhydrazone FCCP or 50 μM dinitrophenol (DNP) and several biochemical assays and techniques of microscopy were used to monitor mitochondria uncoupling-induced lipolysis assessed by glycerol release. | |||
Mitochondrial uncoupling-induces lipolysis but does not involve lipolytic enzymes such as hormone-sensitive lipase (HSL) and adipose ATGL [2]. Enhanced lipolysis relies on a form of autophagy as lipid droplets are directly captured by endolysosomal vesicles. In addition, lysosomal poisoning and inhibition of microautophagy by valinomycin inhibit lipolysis. | |||
A new mechanism of triacylglycerol breakdown was identified in adipocytes exposed to mild uncoupling that provides new insights on the biology of adipocytes dealing with mitochondria forced to dissipate energy. | |||
|editor=[[Plangger M]], [[Kandolf G]] | |editor=[[Plangger M]], [[Kandolf G]] | ||
}} | }} | ||
{{Labeling}} | {{Labeling | ||
|diseases=Obesity | |||
|organism=Mouse | |||
|tissues=Fat, Fibroblast | |||
|preparations=Intact cells | |||
|topics=Uncoupler | |||
|couplingstates=ET | |||
}} | |||
== Affiliations == | == Affiliations == | ||
Arnould T(1), Demine S(4), Tejerina S(5), Bihin B(1), Thiry M(2), Reddy N(1), Raes M(1), Renard P(1) and Jadot M(3) | |||
:::#URBC-NARILIS, Univ Namur | |||
:::#Lab Cell Biology, GIGA-R, Univ Liège | |||
:::#URPhyM-NARILIS, Univ Namur | |||
:::#Center Diabetes Research, Univ Libre Bruxelles; Belgium | |||
:::#EAT-Eppendorf Array Technology. - [email protected] | |||
== Figures == | |||
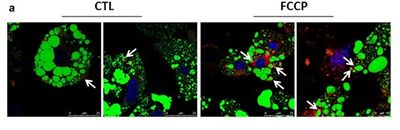
[[File:Arnould_Figure_b_MiP2018.jpg|left|400 px]] Figure 1. Mitochondrial uncoupling increases co-localization between lysosomes and lipid droplets. The co-localization between LDs and lysosomes was visualized by confocal microscopy in cells in which lysosomes, LDs, and nuclei were stained by LysoTracker Red (red), BODIPY 493/503 (green) and TO-PRO3 iodide (blue), respectively. White arrows indicate co-localization events between LDs and lysosomes. | |||
== References == | == References == | ||
::::#De Pauw A, Tejerina S, Raes M, Keijer J, Arnould T (2009) Mitochondrial (dys)function in adipocyte (de)differentiation and systemic alterations. Am J Pathol 175:927-39. | |||
::::#Demine S, Tejerina S, Bihin B, Thiry M, Reddy N, Renard P, Raes M, Jadot M, Arnould T (2018) Mild mitochondrial uncoupling induces HSL/ATGL-independent lipolysis relying on a form of autophagy in 3T3-L1 adipocytes. J Cell Physiol 233:1247-65. | |||
Revision as of 10:06, 20 August 2018
| A form of autophagy triggers lipolysis in 3T3-L1 adipocytes exposed to a mitochondrial uncoupling. |
Link: MiP2018
Arnould T, Demine S, Tejerina S, Bihin B, Thiry M, Reddy N, Raes M, Renard P, Jadot M (2018)
Event: MiP2018
Obesity is characterized by an excessive triacylglycerol accumulation in white adipocytes. Various mechanisms allowing the regulation of triacylglycerol storage and mobilization by lipid droplet-associated proteins as well as lipolytic enzymes have been identified. Increasing energy expenditure by inducing a mild uncoupling of mitochondria in adipocytes might represent a putative interesting anti-obesity strategy [1] as it reduces the adipose tissue triacylglycerol content by stimulating lipolysis through yet unknown mechanisms, limiting the systemic adverse effects of adipocyte hypertrophy.
3T3-L1 fibroblasts were exposed to a mild uncoupling of mitochondria triggered by 0.5 μM carbonyl cyanide-p-trifluoromethoxyphenylhydrazone FCCP or 50 μM dinitrophenol (DNP) and several biochemical assays and techniques of microscopy were used to monitor mitochondria uncoupling-induced lipolysis assessed by glycerol release.
Mitochondrial uncoupling-induces lipolysis but does not involve lipolytic enzymes such as hormone-sensitive lipase (HSL) and adipose ATGL [2]. Enhanced lipolysis relies on a form of autophagy as lipid droplets are directly captured by endolysosomal vesicles. In addition, lysosomal poisoning and inhibition of microautophagy by valinomycin inhibit lipolysis.
A new mechanism of triacylglycerol breakdown was identified in adipocytes exposed to mild uncoupling that provides new insights on the biology of adipocytes dealing with mitochondria forced to dissipate energy.
• Bioblast editor: Plangger M, Kandolf G
Labels: Pathology: Obesity
Organism: Mouse Tissue;cell: Fat, Fibroblast Preparation: Intact cells
Regulation: Uncoupler Coupling state: ET
Affiliations
Arnould T(1), Demine S(4), Tejerina S(5), Bihin B(1), Thiry M(2), Reddy N(1), Raes M(1), Renard P(1) and Jadot M(3)
- URBC-NARILIS, Univ Namur
- Lab Cell Biology, GIGA-R, Univ Liège
- URPhyM-NARILIS, Univ Namur
- Center Diabetes Research, Univ Libre Bruxelles; Belgium
- EAT-Eppendorf Array Technology. - [email protected]
Figures
Figure 1. Mitochondrial uncoupling increases co-localization between lysosomes and lipid droplets. The co-localization between LDs and lysosomes was visualized by confocal microscopy in cells in which lysosomes, LDs, and nuclei were stained by LysoTracker Red (red), BODIPY 493/503 (green) and TO-PRO3 iodide (blue), respectively. White arrows indicate co-localization events between LDs and lysosomes.
References
- De Pauw A, Tejerina S, Raes M, Keijer J, Arnould T (2009) Mitochondrial (dys)function in adipocyte (de)differentiation and systemic alterations. Am J Pathol 175:927-39.
- Demine S, Tejerina S, Bihin B, Thiry M, Reddy N, Renard P, Raes M, Jadot M, Arnould T (2018) Mild mitochondrial uncoupling induces HSL/ATGL-independent lipolysis relying on a form of autophagy in 3T3-L1 adipocytes. J Cell Physiol 233:1247-65.