DTPA: Difference between revisions
From Bioblast
No edit summary |
No edit summary |
||
| Line 3: | Line 3: | ||
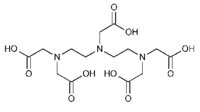
|description='''DTPA''' (Diethylenetriamine-N,N,N',N,N-pentaacetic acid, pentetic acid,(Carboxymethyl)imino]bis(ethylenenitrilo)-tetra-acetic acid) is a polyaminopolycarboxylic acid (like EDTA) chelator of metal cations. DTPA wraps around a metal ion by forming up to eight bounds, because each COO- group and and N-center serves a center for chelation. With transition metals the number of bounds is less than eight. The compound is not cell membrane permeable. In general, it chelates multivalent ions stronger than EDTA. | |description='''DTPA''' (Diethylenetriamine-N,N,N',N,N-pentaacetic acid, pentetic acid,(Carboxymethyl)imino]bis(ethylenenitrilo)-tetra-acetic acid) is a polyaminopolycarboxylic acid (like EDTA) chelator of metal cations. DTPA wraps around a metal ion by forming up to eight bounds, because each COO- group and and N-center serves a center for chelation. With transition metals the number of bounds is less than eight. The compound is not cell membrane permeable. In general, it chelates multivalent ions stronger than EDTA. | ||
}} | }} | ||
[[File:DTPA structure.gif|right|200px|DTPA structure]] | |||
__TOC__ | __TOC__ | ||
== Application in [[HRR]] == | == Application in [[HRR]] == | ||
| Line 49: | Line 45: | ||
::::* Stock solution: 5 mM | ::::* Stock solution: 5 mM | ||
::::* Titration volume: 6 µL | ::::* Titration volume: 6 µL | ||
{{MitoPedia concepts | {{MitoPedia concepts | ||
Revision as of 15:48, 16 March 2021
Description
[[Description::DTPA (Diethylenetriamine-N,N,N',N,N-pentaacetic acid, pentetic acid,(Carboxymethyl)imino]bis(ethylenenitrilo)-tetra-acetic acid) is a polyaminopolycarboxylic acid (like EDTA) chelator of metal cations. DTPA wraps around a metal ion by forming up to eight bounds, because each COO- group and and N-center serves a center for chelation. With transition metals the number of bounds is less than eight. The compound is not cell membrane permeable. In general, it chelates multivalent ions stronger than EDTA.]]
Abbreviation: DTPA
Application in HRR
- Fluorometry:
- In Amplex UltraRed assay systems with HRP, there is a spontaneous increase in the UltoroxRed fluorescence signal over the experimental time without any biological sample. This high background flux can be related to iron contamination. We have done a set of experiments, where we added DTPA as an iron-chelator into the O2k-chamber during AmR measurements. As a matter of fact, DTPA significantly reduced the high chemical background flux in MiR05 without affecting respiration.
- Administration of DTPA into the O2k-chamber is recommended before all other chemicals because the iron chelation of the compound is time-dependent; it takes 10-15 min.
- Experiments may also be run without DTPA.
- DTPA (C2H2O2); Dr.Ehrenstorfer GmbH (E 13095000) or Sigma Aldrich (D-1133); CAS 67-43-6; The commercial powder can be stored at room temperature. The stock solution has to be stored at -20 °C; M: 393.35 g·mol-1
- Fluorometry:
- Solubility: It is soluble in water (4,8 g/l at 25 °C), but not readily soluble in ethanol or apolar solvents.
- Stability constant: see: [1]:
Metal DTPA EDTA Ca(II) 10.74 10.96 Mg(II) 9.3 8.69 Fe(II) 16.5 14.3 Fe(III) 28.6 25.1 Mn(II) 15.6 14.04 Zn(II) 18.75 16.5
- Preparation of the 5 mM stock solution of DTPA:
- Weigh 1.967 mg of DTPA in a 2 mL glass vial.
- Dissolve in 500 µL ddH2O and add 6 µL of 5M KOH to set the pH.
- Heat up the solution up to 33 °C.
- Add 2 µL 5 M KOH to adjust the pH ~ 8-9.
- Add 500 µL ddH2O to get 5 mM stock solution.
- The solution has to be prepared in glass vials, but after then it can be stored in Eppendorf tubes at -20 °C.
- »O2k manual titrations: MiPNet09.12 O2k-Titrations
- Final concentration in O2k-chamber: 15 µM
- Stock solution: 5 mM
- Titration volume: 6 µL
- »O2k manual titrations: MiPNet09.12 O2k-Titrations
MitoPedia concepts:
Recommended
MitoPedia methods:
Fluorometry